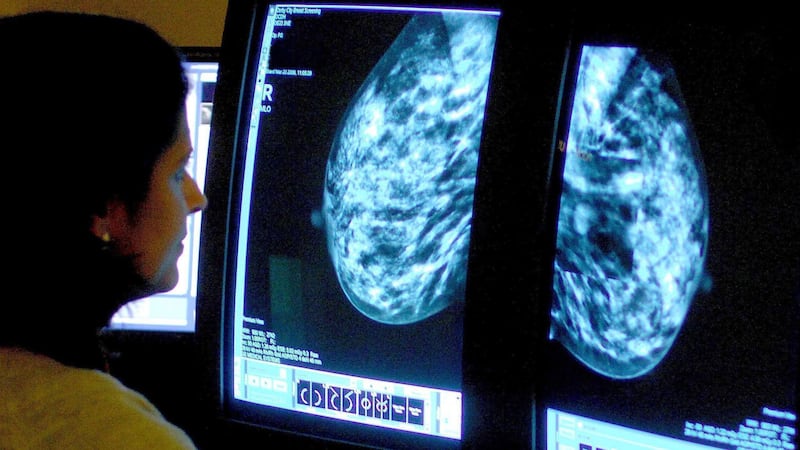
A “landmark moment” in the treatment of advanced breast cancer could benefit around 8,000 women in the UK each year, scientists have said.
A new targeted drug has shown “fantastic” results – shrinking tumours and doubling the amount of time people have before their disease progresses.
A phase III clinical trial of capivasertib alongside hormone therapy suggests the combination could become the new treatment for patients with advanced forms of the most common type of breast cancer.
The findings, presented at the San Antonio Breast Cancer Symposium on Thursday, relate to oestrogen receptor (ER) positive, human epidermal growth factor receptor 2 (HER-2) negative breast cancer, which accounts for around 70% of all new breast cancer cases.
Capivasertib is being manufactured by pharmaceutical giant AstraZeneca following a programme of drug discovery research at the Institute of Cancer Research (ICR) in London.
The drug’s novel action blocks activity of the cancer-driving protein molecule AKT.
All the men and women on the trial, led by researchers at the ICR and the Royal Marsden NHS Foundation Trust, had seen their cancer recur or progress on standard hormone treatments, and the majority had also previously been treated with CDK4/6 inhibitors – drugs that block cancer cells from multiplying.
At present, patients whose cancer progresses continue to receive fulvestrant hormone therapy, but this is often not effective, and many are left only with the option of chemotherapy.
For the research, scientists randomly assigned 355 patients to receive capivasertib plus fulvestrant and 353 patients to receive a placebo plus fulvestrant.
The results showed that treatment with capivasertib plus fulvestrant typically gave patients 7.2 months without their disease progressing or getting worse, compared to 3.6 months for patients treated with placebo plus fulvestrant.
The treatment shrank tumours in 23% of patients, compared with 12% of patients who received fulvestrant plus a placebo.
The new drug regime was also more effective for the four in 10 patients whose cancers had mutations to the AKT signalling pathway, with 29% seeing their tumours shrink compared to 10% on the placebo combination.
Genetic alterations to the AKT pathway can drive both cancer’s development and resistance to treatment.
Trial leader Nick Turner, professor of molecular oncology at the ICR and consultant oncologist at the Royal Marsden, said: “This is a fantastic finding for patients with breast cancer.
“Even with the best current treatments, people with this type of advanced breast cancer will eventually see their cancer stop responding to treatment, and it will progress.
“We’re delighted that this potential first-in-class drug combined with hormone therapy can lower the progression of these advanced cancers, and in almost a third of cases can shrink tumours.
“We believe this new treatment could allow more women and men to live well and live longer with breast cancer.”
Professor Kristian Helin, chief executive of the ICR, said: “This is a landmark moment for the treatment of advanced forms of the most common type of breast cancer.
“It’s incredibly exciting to see a drug that was discovered following research conducted at the ICR now show remarkable benefits for patients in a phase III trial.
“Capivasertib could offer a completely new treatment option for these patients.
“This is a major success story for UK science – the discovery and development of capivasertib showcases the benefits of collaboration between academia, charities and industry to bring game-changing new treatments to people with cancer as quickly as possible.”
Prof Helin said he hoped longer-term follow up of results would show that patients lived longer on the new drug combination.
“But the existing findings are already strong enough for capivasertib to be submitted to regulators to be considered for approval as a new breast cancer treatment,” he said.
Researchers in the ICR have worked for several years on the science behind getting such a drug to work, including how it would target AKT.
The drug was then developed by AstraZeneca with a view to potentially using it on several cancers.
Linda Kelly, 65, from Milton Keynes, joined the trial at the Royal Marsden in August 2021 after her breast cancer spread to her bones and chest wall.
She said: “The results have been amazing. There has been a substantial reduction in my disease, my cancer has not progressed, and I haven’t had any new tumours.
“The treatment is far less debilitating than chemotherapy and I’ve been given the gift of a longer life.”