For millions of failed dieters, the arrival of Wegovy — a ‘miracle’ treatment offering a guaranteed slimming effect — seems like the discovery of the Holy Grail.
Around two-thirds of us are living with obesity or unhealthy extra weight, with one in four children, teenagers or young adults affected.
As well as immediate problems such as arthritis as a result of carrying excess weight, there are serious long-term complications of obesity — heart disease, cancer and a risk of type 2 diabetes (women with a body mass index, or BMI, of 35 are up to 90 times more likely to have type 2 than someone of a healthy weight, according to a study last year by Aberdeen University).
Clinical trials of weekly injections of semaglutide (Wegovy is the brand name) found that people could lose up to 20 per cent of their body weight in 17 months. The Step 1 studies involved 1,961 severely obese patients from around the world, including the UK.
Two-thirds of the group received weekly injections of semaglutide, while the remainder had dummy injections. All went on a slightly lower-calorie diet and followed a suggested exercise programme of two hours of weekly walks, as well as undergoing monthly counselling sessions. None of those involved knew whether they were receiving the active drug.
At the end of the trial, the semaglutide group had lost an average of almost 34lb (15 kg), with considerable improvements in heart disease risk factors such as blood pressure (the control group lost just 5.7lb, or 2.6 kg).

Other studies, called Step 2 to Step 8 trials, have shown significant weight loss in 20 weeks and dramatic reductions in the risk of type 2 diabetes.
Earlier this month the National Institute for Health and Care Excellence (Nice) recommended that semaglutide should be made available on the NHS, initially in England, for two years through specialist weight-loss management centres, to people with a BMI of 35 and at least one weight-related condition such as high blood pressure (or, exceptionally, to those with a BMI of 30-34.9).
The Nice guidance also said semaglutide should be prescribed alongside a reduced-calorie diet and increased activity.
Around a quarter of us fir into the obesity category with a BMI of 30 and above and overall almost 70 per cent of us are too heavy, while the population is getting steadily larger.
With such dramatic trial results, it is unsurprising that semaglutide and a stream of similar weight-loss drugs in the pipeline are being seen as offering real hope for many living with obesity.
It has also been reported that government officials are drawing up plans for drug companies to bid for multi-billion-pound contracts to provide the treatment much more widely.
Semaglutide was first developed more than 10 years ago by the Danish pharma company Novo Nordisk to treat type 2 diabetes. Marketed as Ozempic, it is already available on NHS prescription (although at slightly different, usually lower, doses to Wegovy).
Studies in patients with diabetes showed it also led to weight loss, raising the question of whether it would benefit people living with obesity who had not yet developed type 2 diabetes.
Semaglutide works by mimicking the effect of a hormone in our bodies called glucagon-like peptide-1 (GLP-1), which is released from the intestine after eating to signal fullness. The drug therefore reduces excess appetite and stops people feeling hungry.
It also slows down stomach emptying and digestion; stimulates the production of insulin, a hormone that removes excess sugar in the blood after eating; and reduces production of another natural hormone called glucagon, which is also involved in blood sugar regulation. These effects curb the symptoms and damage caused by type 2 diabetes.
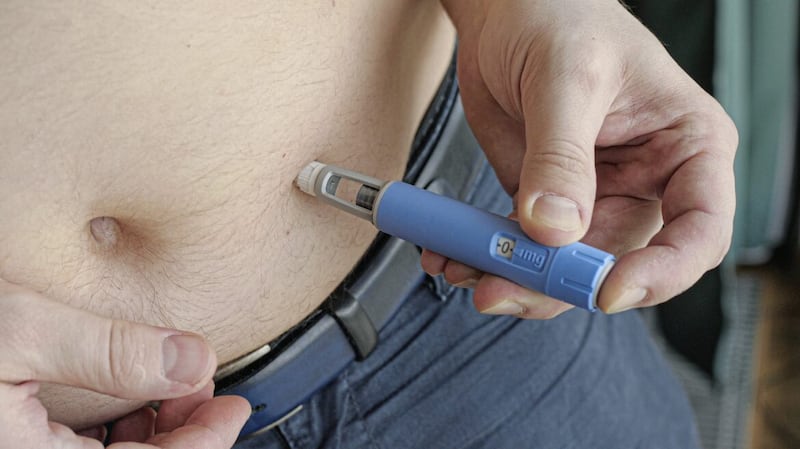
Wegovy is administered by a weekly injection into the stomach with a pre-loaded pen (which users do themselves), with doses increasing progressively until you reach the full dose — and is already available privately, with prices starting at £73 a month for those who can afford it.
These reportedly include Kim Kardashian, who is said to have used it to squeeze herself into a dress worn by Marilyn Monroe for last year’s Met Gala in New York.
The space billionaire Elon Musk said he’d shed weight using it (alongside fasting); while Jeremy Clarkson has also admitted he is taking semaglutide, which he had ordered online.
For everyone else, despite the recent Nice recommendation, the chance of an NHS prescription remains slim. That’s because there has been a surge in people seeking it for weight loss, leading to a worldwide semaglutide shortage.

Nice had actually drafted its semaglutide approval guidance in June last year, but delayed publication because Novo Nordisk could not meet the demand until it had expanded its production.
Tam Fry, a spokesman for the National Obesity Forum, said he has no doubt semaglutide is a game-changer, but that "word has got round that it’s a fantastic drug which will take pounds off you".
Yet "Novo Nordisk hasn’t really taken notice of that and only manufactured a relatively small batch", he adds. "It will mean morbidly obese people who really need it — and for whom it’s a real lifesaver — won’t be able to get it." The drug’s popularity following celebrity use is not the only factor limiting access to it.
Ahmed Ahmed, a bariatric surgeon and clinical senior lecturer in bariatric surgery at Imperial College London, says only half the UK patient population has access to the specialist weight-management centres that can prescribe the drug on the NHS, and waiting lists can be longer than a year.
"At the moment there’s very high demand from people and no supply," he says. "Nice has been very clever; it has made semaglutide hard to get.
"If it made it possible for GPs to prescribe then demand would be through the roof.
"Some [doctors] will be sceptical anyway. There has always been inherent prejudice and stigmatisation of obesity. Half the medical profession don’t agree it’s a disease."
And while he says semaglutide offers genuine benefits, he believes drugs will not provide the answer to our obesity crisis.
"They only really produce a weight loss of 5 or 10 per cent in practice, even if the trials showed much better results," he says.
"If you have someone with a BMI of 35 to 40, then a weight loss of 5 or 10 per cent will be quite good for their diabetes but not enough to ease their arthritic knee pain.
"You can get 30 per cent weight loss with bariatric [i.e. weight-loss] surgery — that’s still the most effective treatment and we need to focus on offering it more widely."
However, others disagree.
"We are entering a new era in which effective and safe drugs for treating obesity are available," said Nick Finer, an honorary clinical professor at the National Centre for Cardiovascular Prevention and Outcomes at University College London (who has previously worked for Novo Nordisk).
"Overwhelming evidence over decades shows that with current approaches few people can lose weight and maintain weight loss.
"Results from another semaglutide trial involving 17,000 people and looking at its impact on reducing heart disease, stroke and death are due out later this year."
He adds: "Real proof of the value of the drug will be when it is shown that this weight loss reduces obesity complications. We know that the same drug in lower doses for people with [type 2] diabetes reduces mortality, stroke and heart attack risk, so hopefully this will also translate to people with obesity."
With obesity expected to affect half of the global population aged over five by 2035, drug market analysts predict that the world market for GLP-1-type drugs (such as semaglutide) will soon be worth more than the £155 billion spent every year on cancer treatments. Another pharma giant, Eli Lilly, is poised this summer to launch a rival to semaglutide: tirzepatide, branded as Mounjaro, which seems to have produced even better weight-loss results in trials.
Nice is reviewing this evidence and is due to publish its conclusions in July. Excited investment analysts from the bank UBS have described tirzepatide as "the biggest drug ever".
Other experts point out that numerous anti-obesity drugs have exploded on to the market, only to be quietly withdrawn in the face of major side-effects when they have been given to large numbers of people.
These effects have included, at best, uncontrollable diarrhoea and, with some drugs, strokes, heart attacks and death.
An appetite suppressant drug called sibutramine (branded as Reductil) was associated with increased risk of stroke — and it was withdrawn in 2010; the use of fenfluramine, an amphetamine derivative, for weight loss was stopped in the late 1990s after it was linked to heart-valve damage.
Another slimming drug called rimonabant was withdrawn in 2008, two years after it was launched, because of potential psychiatric side-effects including suicidal thoughts.
Even the weight-loss drugs that are safe and still in use, such as orlistat, are not without their own problems, with unpleasant side-effects such as "oily discharge from the anus".
In animal studies, semaglutide has been linked to thyroid cancer — but there is no data on whether this risk might extend to humans.
Semaglutide has only been available for two years, which is partly why Nice has recommended to limit its use to the same period.
"No data exists on the longer-term impacts of taking this drug," warns Dr Simon Cork, a senior lecturer in physiology at Anglia Ruskin University.
Another question is what happens once you stop taking it. For instance, a study in the Journal of Pharmacology and Therapeutics found that a majority of people who take semaglutide gain most of the weight back within a year of stopping it.
Dr Cork said: "A follow-up to the initial clinical trial, where participants were monitored after stopping semaglutide, showed that all regained almost all of the weight they had lost over the course of the following year.
"This demonstrates the fact that obesity is a lifelong condition and that semaglutide is a treatment rather than a cure."

"The history of previous weight-loss drugs reveals serious side-effects — from heart problems to suicide — that were concealed," adds David Healy, a professor of psychiatry at Bangor University and author of Pharmageddon, which exposes conflicts of interest between drug companies and researchers working for them.
"We are left to hope that clinical use will show this new drug works for some and is relatively free of serious problems. It would be best for both doctors and patients to approach its use judiciously until we know more, but the lure of the marketing may make waiting to see a difficult stance to take." There is no suggestion that side-effects of Wegovy have been concealed.
Chris van Tulleken, an associate professor at University College London, who has written books highlighting the link between the rise of obesity and our increasingly unhealthy diet, is also to be convinced by semaglutide.
"We know it won’t be nearly as good as the drug company says it is because no drug in history has ever lived up to the promise of the headlines," he says.
"It won’t be a cure for obesity for most people who take it; it’s just one of the best tools we have at the moment. It is also interfering with a very fundamental part of the brain that gets reward from food and fullness.
"What concerns me is that we haven’t been able to focus on prevention. We know the primary cause of pandemic obesity is ultra-processed food — predatory corporations selling addictive food that does enormous harm to people’s bodies, especially children, and we have to limit the marketing of that food.
"It’s great to have a new drug we can spend billions on, but we need to match that with at least as much on prevention."
A Novo Nordisk spokesman said: "We continuously collect and analyse data on the use of our medicines post-marketing authorisation, and follow international pharmacovigilance standards to report and analyse any adverse events experienced by people taking our medicines.
"We work closely with the Medicines and Healthcare products Regulatory Agency to ensure that healthcare professionals have a thorough and full understanding of the safety profile of our medicines."
© Solo dmg media